Quanta-S Steam Sterilizable Freeze Dryers
We explore all design and budget considerations to help meet your pharmaceutical freeze drying requirements with the best possible equipment match.
Our knowledgeable engineers and consultants will help you explore all aspects of your pharmaceutical freeze dryer requirement to help ensure you get the right system for your purpose.
Some options for consideration include:
- multiple skids for tight installation configurations,
- redundant safety systems
- clean-in-place (CIP)
- isolation valves
- automatic or hand secured door latches
- round vs square chamber configuration
- internal vs external condensers
- stoppering or bulk
- bellow seals
- level loading doors
- pass through design
- a variety of documentation and testing
Pharmaceutical Freeze Drying in a Sterile EnvironmentSterile production environments are most commonly found in bio-pharmaceutical and pharmaceutical organizations.
Millrock Technology, Inc supplies cGMP pharmaceutical freeze dryers with steam sterilization and 21CFR part 11 for these demanding applications.
Our support lasts throughout the pharmaceutical freeze dryer life cycle to help ensure that your project starts successfully and remains successful.
Our due diligence helps identify every consideration for a successful installation.
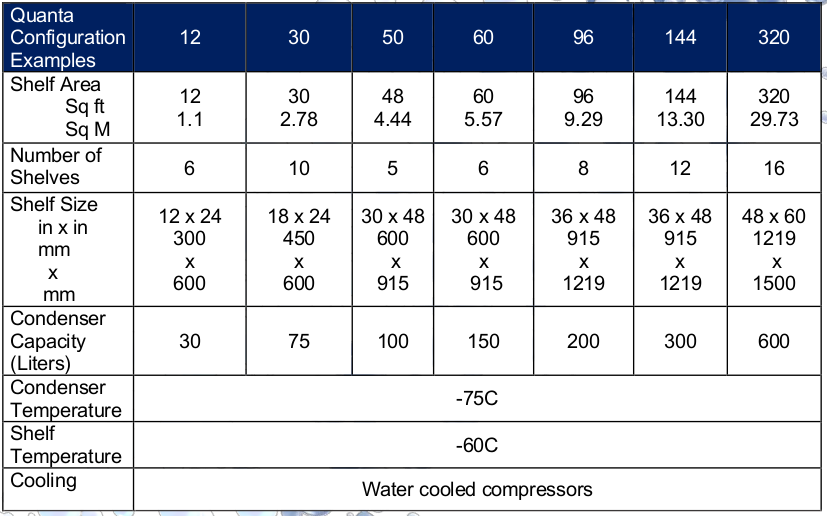
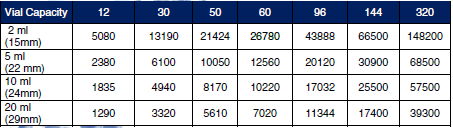
Lyophilizer Sterilization for Commercial Product Freeze DryingOur Steam Sterilizable Production Freeze Dryers include:
- All wetted components 316L stainless steel
- Sterilization temperature: Controllable 121-125 °C
- Evacuation and drying: Water ring pump
- Insulation: High temperature CFC-Free foam with a stainless steel shroud around both chambers
- Filter: 0.2 micron Sartorius with integrity testing ports
- System cooling to ambient and drying in less than 2 hours using the refrigeration system and a liquid ring pump
- Slope to drain